The internal and external genitalia of both sexes are genetically ‘programmed’ to differentiate passively down a ‘default’ female pathway unless actively switched down a male pathway by the genetic information carried by the testis-determining gene (SRY). Until the sixth week of gestation, the genitalia of both sexes share identical embryonic precursors. Differentiation of the gonads and genital tracts is initiated by the migration of primordial germ cells from the yolk sac, across the coelomic cavity to condensations of primitive mesenchyme in the lumbar region of the embryo.
By a process of reciprocal interaction, the germ cells and surrounding mesenchyme form the primitive sex cords within the embryonic gonad.
At around this time (6 weeks), the paired paramesonephric ducts appear as cords of coelomic epithelium lying lateral to the mesonephric ducts. From this stage onwards, the pathways of male and female differentiation diverge.
Female
Internal genitalia
Although the primitive sex cords degenerate, secondary sex cords derived from genital ridge mesoderm enfold the primordial germ cells to form primitive follicles. Differentiation of the genitalia down a female pathway cannot be entirely explained by the absence of the SRY gene. Normal development of the ovary does appear to at least be partly dependent on the presence of the two normal X chromosomes, since patients with Turner syndrome who have a single X chromosome (X karyotype) typically have poorly formed, dysgenetic ovaries.
In the female, the initial phase of gametogenesis (transition from primordial germ cell to primary oocyte) occurs within the fetal ovaries.
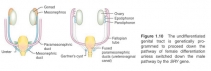 Figure 1.10 The undifferentiated genital tract is genetically programmed to proceed down the pathway of female differentiation unless switched down the male pathway by the SRY gene.
Figure 1.10 The undifferentiated genital tract is genetically programmed to proceed down the pathway of female differentiation unless switched down the male pathway by the SRY gene.
During fetal life these primary oocytes embark on the first phase of meiotic division before entering a long phase of arrested division, which resumes again only at puberty. In the absence of testosterone, the mesonephric ducts regress (leaving only vestigial remnants – the epoöphoron, paroöphoron and Gartner’s cysts). The paramesonephric ducts persist in the form of the fallopian tubes. Distally, the fused portions of the paramesonephric ducts give rise to the uterus and upper two-thirds of the vagina.
At the junction of the paired paramesonephric ducts with the urogenital sinus a condensation of tissue, the sinuvaginal bulb, develops. Between the 10th and 20th weeks of gestation displacement of the sinuvaginal bulb in the direction of the fetal perineum separates the developing vagina from the urethra. During this process, canalisation of the vagina occurs. The upper two-thirds of the vagina is derived from the paramesonephric ducts, whereas the distal third has its origins in the urogenital sinus and the introitus and external genitalia are derived from ectoderm.
 Figure 1.11 Development of the lower female genital tract between 10 and 20 weeks.
Figure 1.11 Development of the lower female genital tract between 10 and 20 weeks.
External genitalia
In the absence of androgenic stimulation, the external genitalia of the embryo and fetus differentiate passively down a female pathway. The genital tubercle gives rise to the clitoris, the urogenital sinus contributes the vestibule of the vagina, while the urogenital folds persist as the labia minora, and the labioscrotal folds persist as the labia majora.
{pagebreak}
Male
Internal genitalia
Current evidence indicates that differentiation of the male genitalia is initiated by a single testisdetermining gene (SRY) located on the Y chromosome and then mediated through other Y chromosomal and autosomal ‘downstream’ genes.
The gene product expressed by the SRY gene is responsible for stimulating the medullary sex cords to differentiate into secretory pre-Sertoli cells.
From the seventh week onwards these pre-Sertoli cells secrete anti-mûllerian hormone (AMH) – otherwise termed mûllerian inhibiting substance (MIS), a glycoprotein that plays a central role in subsequent differentiation of the male genital tract.
In the male the paramesonephric ducts disappear completely, with the exception of vestigial remnants (the appendix testis and utriculus).
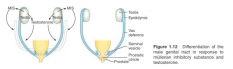 Figure 1.12 Differentiation of the male genital tract in response to mûllerian inhibitory substance and testosterone.
Figure 1.12 Differentiation of the male genital tract in response to mûllerian inhibitory substance and testosterone.
At least three important properties are ascribed to MIS:
1. MIS stimulates regression of the paramesonephric ducts.
2. Production of testosterone by the Leydig cells of the embryonic testis is initiated by MIS from the ninth week of gestation. During the 12th to 14th weeks of gestation the fetus is exposed to very high levels of androgenic stimulation.
3. The first stage of testicular descent is mediated by the action of MIS on the gubernaculum, which anchors the embryonic testis in the vicinity of the developing inguinal canal.
 Figure 1.13 Differentiation of the external genitalia determined by androgenic stimulation.
Figure 1.13 Differentiation of the external genitalia determined by androgenic stimulation.
In contrast to the germ cell differentiation initiated in the female embryo, male primordial germ cells are inhibited from further division within the embryonic gonad. Although gametogenesis does not commence until after puberty the testis is not entirely quiescent in childhood since there is evidence that the fetal gonocyte germ cell pool transforms into adult type gonocytes in early childhood. The mesonephric duct derivatives differentiate between the eighth and 12th weeks of gestation to give rise to the epididymis, rete testis, vas deferens, ejaculatory ducts and seminal vesicles. This occurs in response to exposure to testosterone (probably by diffusion) from the developing testis.
The development of the prostate gland is dependent upon circulating testosterone and provides a further example of reciprocal induction.
Proliferation and branching of the endodermal lining of the urethra (which gives rise to the ducts and glanular acini of the prostate gland) induces differentiation of surrounding mesenchyme to form the capsule and smooth muscle, a process that ceases after the 15th week of gestation.
{pagebreak}
External genitalia
A number of factors are responsible for the differentiation of the male external genitalia, including the synthesis of testosterone by the fetal testis, the conversion of testosterone into dihydrotestosterone by the enzyme 5a-reductase and the presence of androgen receptors within the target cells. Androgenic stimulation of the genital tubercle results in the development of the male phallus.
From the seventh week of gestation the male urogenital sinus advances on to the phallus as the urethral groove. Ingrowth of this urethral groove is associated with the appearance of urethral plate tissue destined to canalise and form the definitive male anterior urethra. Closure of the urethra is complete by around 15 weeks, with ingrowth of ectoderm from the tip of the glans forming the terminal portion of the urethra.
The testis
Tesicular descent is believed to occur in two distinct phases, the first initiated by MIS, the second being dependent upon exposure to testosterone.
Experimental studies in rats have indicated that the gubernaculum plays a key role in testicular descent (although the precise mechanism may be subject to some species variation). Extending from the testis down to the region of the labioscrotal swellings, the gubernaculum has the effect of anchoring the fetal testis in the proximity of the future inguinal canal. Under the influence of testosterone, a second more active phase of testicular descent occurs between 25 and 30 weeks, when the gubernaculum contracts in length, dragging the testis down the inguinal canal into the scrotal position it occupies at birth. During the course of its descent the testis is accompanied by a sac-like protrusion of peritoneum, the processus vaginalis.
Clinical considerations
Abnormalities of female internal genitalia
Patterns of faulty development of the paramesonephric duct derivatives include agenesis, hypoplasia, defective canalisation and duplication.
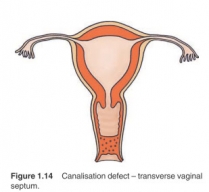 Figure 1.14 Canalisation defect – transverse vaginal septum.
Figure 1.14 Canalisation defect – transverse vaginal septum.
Unilateral agenesis of paramesonephric duct derivatives (absent fallopian tube, hemiuterus, etc.) may be accompanied by unilateral renal agenesis, suggesting a fundamental underlying defect of the original ipsilateral intermediate mesoderm.
Agenesis of the upper two-thirds of the vagina (Rokitansky syndrome) reflects a failure of the paramesonephric ducts to fuse distally and merge with the urogenital sinus. Transverse vaginal septa and short atretic segments can be attributed to defective canalisation (Figure 1.14), whereas duplication anomalies such as bicornuate uterus represent incomplete fusion of the distal paramesonephric ducts (Figure 1.15).
 Figure 1.15 Fusion defect – bicornuate uterus.
Figure 1.15 Fusion defect – bicornuate uterus.
Female external genitalia
The aetiology and classification of disorders of sexual development (DSD) are considered in more detail in next Section. The most commonly encountered example in Western Europe is virilisation of the external genitalia in a female with a 46XX genotype resulting from congenital adrenal hyperplasia.
In affected individuals, the internal reproductive tract differentiates normally but the external genitalia virilise in response to high levels of circulating androgens of adrenal origin.
{pagebreak}
Male internal genitalia
The rare genetically determined syndrome of MIS deficiency is characterised by bilateral undescended testes accompanied by the presence of persistent paramesonephric duct structures, including fallopian tubes and uterus. A receptor defect is responsible for approximately 50% of cases, whereas the remaining cases are genuinely attributable to a deficiency of this hormone. With the exception of the undescended testes, the external genitalia virilise normally as this is not dependent on MIS.
The persistence of a mûllerian sac-like prostatic utricle is predominantly a feature of impaired virilisation and patients with proximal hypospadias or DSD account for more than 90% of cases.
In the syndrome of androgen insensitivity, the external genitalia exhibit a receptor defect to dihydrotestosterone (the derivative of testosterone active in the peripheral tissues). As a result, the external genitalia differentiate passively down the female pathway despite a male phenotype and normal male internal genitalia.
Male external genitalia
Hypospadias results from varying degrees of incomplete closure of the urethral groove (although distal glanular hypospadias may simply represent failure of ectodermal ingrowth). Severe hypospadias, particularly when accompanied by cryptorchidism and a persistent mûllerian utriculus, is evidently the outcome of a generalised virilisation defect.
However, although isolated endocrinopathies have occasionally been identified in such cases, as yet no single endocrine defect has been implicated in the aetiology of hypospadias.
Similarly, there is no single unifying aetiology for cryptorchidism. Bilateral cryptorchidism is more likely to represent the influence of an endocrinopathy or imbalance of the pituitary-gonadal axis, whereas local mechanical factors (possibly related to the gubernaculum or the processus vaginalis) are likely to play a more important role in the unilateral cryptorchidism.
The combination of a blind-ending vas and histological findings of calcification and haemosiderin in ‘nubbins’ of residual testicular tissue suggests that most cases of so-called testicular ‘agenesis’ result from testicular torsion in utero.
{pagebreak}
Key points
- The genitourinary tract is commonly affected in children with chromosomal abnormalities.
- The ureteric bud plays a pivotal role in nephrogenesis and the embryological development of the upper tract.
- A number of important congenital anomalies can be ascribed to defects of the ureteric bud.
- The undifferentiated genital tract of both sexes is ‘programmed’ to differentiate passively down a female pathway unless positively directed down a male pathway by the presence of the SRY gene and associated downstream genes and their products.
- Although single gene mutations have been implicated in the aetiology of some inherited genitourinary abnormalities, most of the conditions encountered in paediatric urology occur on a sporadic basis or result from the interaction of multiple genes.
- In vitro fertilisation techniques (IVF) offer the prospect of fertility to some patients whose genitourinary malformations would previously have been a bar to parenthood.
Further reading
Hutson JM, Terada M, Baiyun Z, Williams MPL. Normal testicular descent and aetiology of cryptorchidism. Advances in Anatomy, Embryology and Cell Biology. Berlin: Springer Verlag, 1996: 132
Larsen WJ (ed). Human Embryology. Edinburgh: Churchill Livingstone, 2002
McNeal JE. Anatomy and embryology. In: Fitzpatrtick JM, Kane RJ (eds). The Prostate. Edinburgh: Churchill Livingstone, 1989: 3-9
Stahl DA, Koul HK, Chacko JK, Mingin GC. Congenital anomalies of the kidney and urinary tract (CAKUT): a current review of cell signalling processes in ureteral development. J Pediatr Urol 2006; 2: 2-9
Thomson JA, Itskovitz-Eldor J, Shapiro SS et al. Embryonic stem cell lines derived from human blastocysts. Science 1998; 282: 1145-1147
Woolf AS, Price KL, Scambler PJ, Winyard PJ. Evolving concepts in human renal dysplasia. J Am Soc Nephrol 2004; 15(4): 998-1007

































